|
TITLE: |
|||
|
|
|||
|
DOCUMENT ID: |
3800 Human Subjects
Research (HSR) Protection Program |
||
|
|
|||
1.0
Purpose
Jefferson Lab participates in
conducting joint research and providing technical support in Human
Subjects Research (HSR)
studies. It is the policy of the Laboratory to maintain an agreement by which
HSR is reviewed and approved for the protection of human subjects.
This program has been established
in accordance with 10 CFR 745 Protection of Human Subjects, 45 CFR 46
Protection of Human Subjects, and DOE Order 443.1B Protection of Human Research
Subjects.
A human subject is a living individual about whom an investigator conducting research obtains:
1) Data through intervention or
interaction with the individual, or
2) Personally identifiable information
(PII).
The
requirements of this program apply to any HSR conducted with DOE funding, DOE
institutions regardless of funding, or by DOE contractor personnel regardless
of funding source or location conducted.
No research involving human
subjects can be initiated or conducted without prior approval by the Institutional Review
Board (IRB) that is
registered with the Department of Health and Human Services (DHHS), as
evidenced by a Federal Wide
Assurance (FWA) issued
by the Office of Human
Research Protection (OHRP).
3.1
Principal Investigator
·
Notifies
Procurement Services of proposed work involving HSR (see Figure 1), and requests Cooperative Research and Development
Authority (CRADA) package, or other appropriate agreement.
·
Notify
Director, ES&H if the proposed work involves
o
An
institution without a formal IRB
o
A
foreign country
o
The
potential for significant controversy (e.g., negative press or reaction from
stakeholder or oversight groups)
o
Research
subjects in a protected class (fetuses, pregnant women, and in vitro
fertilization; prisoners; or children
o
The
generation or use of classified information
·
Completes
and submits CRADA, or other agreement, to Procurement Services and other
appropriate parties for review.
·
Provides
FWA number from partner/sponsor as evidence of IRB approval for any contracted
work involving HSR activities.
·
Ensures
compliance with the Human Subjects Protection (HSP)
Program procedures
and other requirements.
·
Immediately
notifies Director, ES&H of:
o
Any
significant adverse event, unanticipated problem, and complaints about the
research;
o
Any
suspension or termination of IRB approval;
o
Any
significant non-compliance with HSP Program procedures or other requirements
which require reporting to the IRB for further evaluation; or
o
Any
suspected or confirmed data breach involving PII in printed or electronic form.
3.2
Director, ES&H
·
Reviews
proposed work involving HSR to assess risks to participants and potential
ES&H hazards involved in research activities.
·
Assures
compliance with applicable Federal and DOE regulations and requirements
involving HSR.
·
Maintains
list of work involving HSR activities.
·
Performs
notifications to DOE HSP Program Manager as required by DOE O 443.1B.
·
Performs
oversight of HSR Protection Program through the Jefferson Lab Contractor
Assurance System (CAS).
·
Ensures
relevant personnel participate in HSP training as appropriate.
3.3
Procurement Services
·
Provides
CRADA package, or other appropriate agreement, to Principal Investigator upon
request.
·
Manages
CRADA process, or other business arrangements, in accordance with department
procedures.
3.4
Everyone at Jefferson Lab
·
Does
not perform any work involving HSR without prior IRB approval.
·
Safeguards
the welfare, privacy, and rights of human subjects who take part in research
experiments.
·
Immediately
notifies Principal Investigator of any unanticipated or potential problems
related or possibly related to HSR activities.
Figure 1
– Does Activity Involve Human Subjects Research?
Modified from
DHHS
Chart 1: Is an Activity Research Involving
Human
Subjects Covered by 45 CFR part 46?
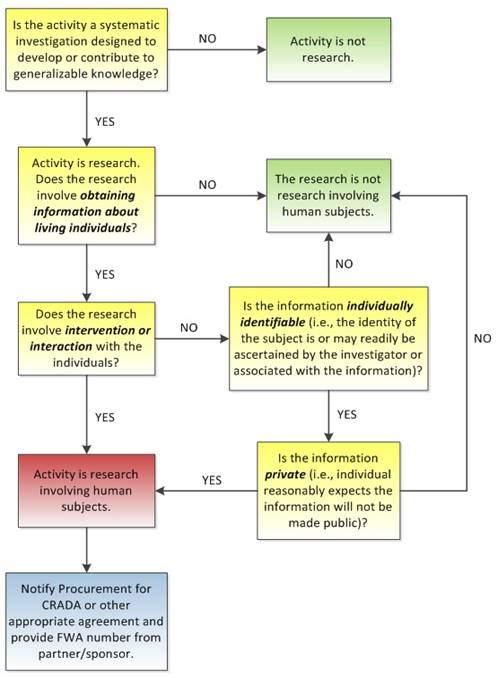
10 CFR 745, Protection of Human
Subjects
45 CFR 46, Protection of Human
Subjects
DOE Order 443.1B, Protection of Human Research Subjects
5.0
Revision
Summary
Revision 1.1 – 06/9/21 – Periodic Review; revised header and footer, changed Associate Director to Director, No approval required.
Revision 1.0 –
06/14/16 – Periodic Review; revised to reflect requirements in DOE
Order 442.1BChg 1.
Revision
0.0 – 07/01/13 – New
material written to reflect current laboratory operations
|
|
ISSUING AUTHORITY |
TECHNICAL
POINT-OF-CONTACT |
APPROVAL
DATE |
REVIEW DATE |
REV. |
|
|
|
ES&H Division |
06/14/16 |
06/9/24 |
1.1 |
|