| ES&H Manual | |||||||||
| Chapter 5200 Event Investigation and Causal Analysis Process | |||||||||
|
Purpose
Jefferson Lab initiates and completes investigations for all notable events, including near misses, and analyzes for Human Performance Improvements (HPI) indicators in an effort to learn from mistakes and gain perspective from the event. This process encourages the development and sharing of lessons learned, corrective actions, and follows the guidelines outlined in DOE Order 232.2A Occurrence Reporting and Processing of Operations Information. All unwanted events reported to the ES&H Reporting Officer are screened to determine if they are "notable," thus subject to a formal investigation, including a causal analysis. This event investigation and causal analysis process enables the Lab to capture, develop, and implement corrective actions to reduce the likelihood of reoccurrence; perform trend analyses, and learn from these unintended events. When deemed necessary, events are reported to DOE in accordance with the following requirements:
NOTE: This process does not negate DOE Order 225.1B Accident Investigation, which allows TJSO to conduct an independent investigation of any event at Jefferson Lab. Any such investigation is coordinated through the Associate Director, ES&H.
Scope
This chapter outlines Jefferson Lab responsibilities to report unwanted events and notify the appropriate personnel. 2.1 Notable Events at Jefferson Lab could include:
2.2 Radiological Events: All radiological events are screened by the ESamp;&H Reporting Officer in conjunction with the Radiation Control (RadCon) Manager in accordance with DOE Order 232.2A Occurrence Reporting and Processing of Operations Information and 10 CFR 835 Occupational Radiation Protection Program. Radiological incidents that do not exceed any relevant reporting criteria, and are not infractions of 10 CFR 835 or other applicable requirements, may be dispositioned in accordance with RadCon standard operating procedures. These incidents shall be documented as "Radiation Deviation Reports" in the Corrective Actions Tracking System (CATS) where they are available for review and trending. 2.3 Other Incidents: Other incidents, including first aid cases, are dispositioned for investigation on a graded approach between the Associate Director, ES&H and the affected Division Safety Officer. The information is compiled and used for safety related trend analysis. In some instances, there are corrective actions associated with the incident that are entered into CATS and tracked to completion. This information is then shared at various meetings with lab management. Any event or trend may be elevated to a Notable Event, or reported through ORPS or NTS as a management concern by the Associate Director, ES&H. Table 1: Required Training Courses for Lead Investigators
|
|||||||||
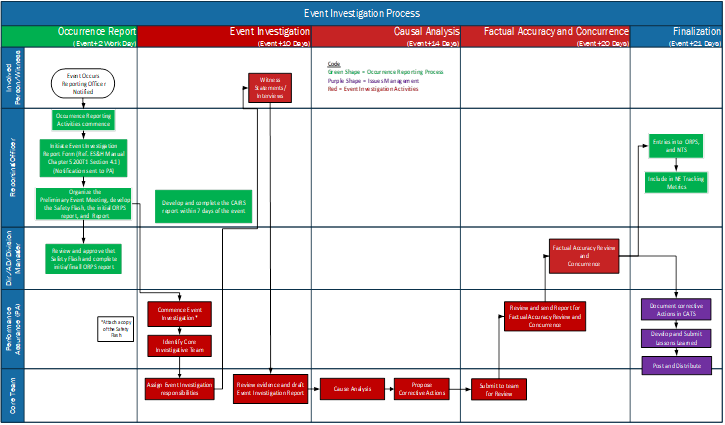
Appendices: 5200T1: Event Investigation and Causal Analysis Process Jefferson Lab notifies the Department of Energy (DOE) - Thomas Jefferson Site Office (TJSO) of any occurrence that could adversely affect the health and safety of the public, its workers, and the environment. Occurrence reporting promotes organizational learning consistent with DOE's Integrated Safety Management System goal of enhancing mission safety and sharing effective practices to support continuous improvement and adaptation to change. 5200T2: Event Investigation and Causal Analysis Procedure This procedure provides the process steps for conducting an event investigation and causal analysis. Steps include a preliminary event assembly, initiation of the event investigation, investigation activities, cause analysis to determine root cause, proposing corrective actions, and submittal for concurrence. Corrective actions are resolved using the Issues and Corrective Action Management Process, and the final report is summarized and distributed within the Lessons Learned Program. References: |
Responsibilities: Note: Management authority may be delegated to a task qualified Jefferson Lab employee at the discretion of the responsible manager. Everyone at Jefferson Lab
Involved Persons
Supervisor/Technical Representative (TR)/Sponsor, Occupational Medicine
ES&H Reporting Officer or designee
ES&H Director
Performance Assurance Manager
Lead Investigator / Core Team Members
|
||||||||
Document Control:
|
|||||||||
|
This document is controlled as an on-line file. It may be printed but the print copy is not a controlled document. It is the user's responsibility to ensure that the document is the same revision as the current on line file. . |
|||||||||