Positron Emission Mammography (PEM)

PEM detectors placed under a patient table. A gelatin breast phantom is positioned between two detectors.
Jefferson Lab is collaborating with Duke University Medical Center, in Durham, NC, the Virginia Commonwealth University Health System in Richmond, VA and the University of West Virginia Medical Center in Morgantown, WV to develop detector technologies based on the imaging of cancer tracers tagged with radioactive isotopes that are used in Positron Emission Tomography (PET). This new medical imaging technique is called Positron Emission Mammography (PEM).
Mammography vs. Positron Emission Mammography (PEM)
Although film screen mammography is currently the best way to detect breast cancer, both the sensitivity and specificity of this technique are limited. The sensitivity of mammography is approximately 88% for detectable breast cancer. The remaining twelve percent are typically detected at physical exam. Moreover, the specificity of mammography is much lower than its sensitivity, approximately 15-35%; thus many women are subjected to breast biopsies for benign lesions. Many of mammography's limits are inherent; mammography utilizes ionizing radiation (x-rays) to produce an image of the breast. This results in an image that reflects the spatial x-ray attenuation of different parts of the breast. Thus the radiologist searches for subtle abnormalities in both attenuation and architecture to identify possible breast cancer. Unfortunately, the attenuation differences between normal fibroglandular tissue and breast cancer are small. Thus, the ability to detect breast cancer is limited in women with radiographically dense breasts. Furthermore, there are many benign masses and calcifications that are indistinguishable from malignant lesions. Finally, some cancers do not produce either attenuation or architectural abnormalities until they are advanced. Mammography is inherently limited in its ability to detect breast cancer because it relies on demonstrating morphologic differences between cancer and benign lesions. Positron Emission Mammography uses injected radioactively "tagged" sugar-like molecules (called FDG) to mark or make visible tissue which is more likely to be cancerous. The cancer tumors accumulate more of the radioactive tracer which appears to the tissue as if it is glucose or sugar, a source of much needed energy for growth. Since tumor cells are growing more quickly than healthy tissue the cancer shows up as "hot" spots in the PEM images.
Positron Emission Tomography (PET)
Positron Emission Tomography (PET) imaging works as follows. A positron-emitting radionuclide labeled to a molecule of interest is injected into the body. This tracer accumulates throughout the body according each tissue type's affinity for the tracer. When a positron is emitted, it travels a short distance (on the millimeter scale) and annihilates with an electron, producing two 511 keV photons, which leave in opposite directions. This anticollinearity is used for localization; when two photons are detected within some small time window, it is assumed that they came from one annihilation, and this annihilation was on the line connecting the detection points. State-of-the-art PET scanners incorporate rings of radiation detectors which surround the body. Thousands of individual detectors are used to achieve maximal spatial resolution, radiation detection efficiency, and count rates.
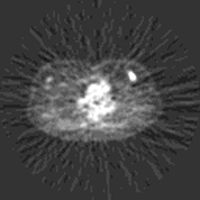
Focal uptake of FDG in the left breast, shown here as a bright spot, indicates malignancy in this study performed at Duke University
PET with the radioactive sugar-like molecule called 2-fluoro-2-deoxy-D-glucose (FDG) produces images that depict the pixel-by-pixel level of metabolic activity in the body. FDG is a cyclotron-produced, positron-emitting glucose analog that goes through the first step of the glycolytic pathway, then becomes intracellularly trapped. The subsequent emission of a positron from each trapped FDG molecule provides a signal for the scanning instrumentation. Malignant cells demonstrate an enhanced rate of glycolysis, even in the presence of oxygen, facilitating their detection by FDG PET imaging. Studies performed on whole body PET scanners have substantiated this technique's ability to identify primary and metastatic breast cancer with higher specificity than conventional techniques, regardless of tissue density. In the following figure a tomographic image showing how well FDG allows for the detection of a malignant tumor in the patient's left breast.
Furthermore, other studies indicate that serial FDG PET imaging permits a noninvasive method for assessing tumor response to therapy. Additionally, PET FDG imaging has provided a sensitive and specific method for evaluating surgical sites to differentiate postoperative scarring from local recurrences. This technique provides unique information based upon biochemical and physiological processes, and is complimentary to that afforded by conventional mammography.
Although these early studies performed on whole body PET scanners were encouraging, they also demonstrated the limits imposed upon whole body imaging techniques. First, they have limited spatial resolution by mammographic standards (intrinsic resolution of ~5mm is degraded to 7-9mm after smoothing to reduce noise). Secondly, the efficiency of ring-based scanners for detecting emitted radiation is limited to that radiation which is emitted in the plane of the rings. Thirdly, most of the photon pairs are lost to attenuation since at least one of the photons must travel through a large amount of tissue (the body) to be detected. Finally, abnormalities demonstrated on these scanners are often difficult to localize, i.e., spatially correlate in three dimensions for purposes of biopsy or treatment. Because of the combined effects of spatial resolution and noise, small lesions are difficult to detect. Moreover, dedicated whole-body PET scanners are expensive and limited to a few centers nationwide.
Several changes have occurred recently that are relevant to the application of FDG in breast imaging. First, the value of and interest in FDG PET for cancer evaluation has lead to the need for commercial FDG distribution sites. Many hospitals are now purchasing FDG from these distribution centers, alleviating the need for an on-site cyclotron. With reimbursement for PET procedures increasing, distribution facilities are likely to flourish. Secondly, specialized positron imaging systems for the breast such as thoses under development at Jefferson Lab have been proposed and built. These systems are either small rings of detectors which surround the breast or planar pairs of detectors located above and below the breast (like the Jefferson Lab system.) These systems have multiple benefits. The cost to build such a device is very low compared to a dedicated whole-body PET scanner (currently $1-$2 million). In addition, the imaging properties are better for the specific task of imaging the breast. Spatial resolution, geometric efficiency for detecting emitted radiation, much lower attenuation and scatter of emitted photons, and the ability to spatially correlate abnormalities with mammographic images of the breast taken with the breast in the same position and level of compression all lead to improved lesion detection and characterization, with lower FDG dose requirements. Ideally these units will enable detection of radiographically occult malignancy (particularly in dense breasts), assist in local staging of patients with known breast cancer, early detection of recurrent cancer, and early assessment of tumor response to radiation and chemotherapy.

